|
Absolute measurements are always difficult. Finding $e/m$ was a scientific feat of its own. You had to know the exact magnetic field and electric field values. Recently this has been slightly changed.įollowing the same lines of reasoning, finding the absolute mass of electrons by experiments was not easy. Recently, from 1960s, the fixed element was an isotope of carbon, called C-12. It was given a fixed mass of 1 (exact), later oxygen was chosen as 16 (exact) and this choice moved back and forth. In the long history of atomic weights, the element with a "fixed" mass was hydrogen. Historically, one of the elements has been given a fixed number as its mass. Learn the Preparation, Uses of Hydrogen, Atomic Mass of Hydrogen, Atomic Number and Its Properties with FAQs. All the atomic weights or atomic masses are relative, relative to an arbitrary element-chosen by humans rather than any fundamental reason. What is Hydrogen - Hydrogen is a highly combustible diatomic gas with the molecular formula H2. Only $1 / 1800$ of that of an atom of hydrogen.īy the way, the absolute mass of atoms in the periodic table is not known. Every hydrogen atom has one proton in its nucleus. previously determined $e / m v^ / 1800$ or the mass of an electron is Hydrogen, at the upper left of the table, has an atomic number of 1. Hydrogen gas forms when two atoms of hydrogen bond together to form a molecule with the formula H2. Very briefly, from a Chapter written by Sir J.J. The molar mass of hydrogen gas is 2.016 grams per mole. The idea that the mass to charge ratio of cathode ray "particle" (modern day electron) is a lot lighter than hydrogen came about indirectly from electrochemical experiments and the concept of equivalents. Thomson already discovered that electron is a lot lighter than a hydrogen atom (about 1800 I believe). 
Using these values gives an atomic mass of 938.8 MeV/ $c^2$ for hydrogen, but the measured atomic mass of hydrogen is 939.0 MeV $/c^2$.Did he know the mass of a hydrogen atom at the time? Because J.J. Proton mass is 938.272 MeV/ $c^2$, electron mass is 511 keV/ $c^2$ and the ionization energy of hydrogen is 14 eV. 
The calculation does not add up, however. the ionization energy of hydrogen is featured. However, as there is only one nucleon in hydrogen, the contributions to B do not feature a any term from nuclear binding, only the binding energy of the single atomic state in hydrogen, i.e. If I take the hydrogen atom, and try to predict its mass this way, I find, unsurprisingly And where B is the net binding energy of a nucleus. 1 mole of C 6H 8O 6 8 moles of hydrogen8 g of hydrogen Thus 1.5 moles of C 6H 8O 6 12 g of hydrogen 1 mole of C 2H 4O 2 4 moles of hydrogen4 g of hydrogen Thus 4 moles of C 3H 8O 3 16 g of hydrogen Thus 4 moles of C 3H 8O 3 contains the largest mass of hydrogen. For example Hydrogen (H) atomic mass -1 Lithium (Li) atomic mass -3 Periodic Table 2. By referring to the periodic table In the periodic table digit of an atomic mass usually marked under the representation of an element. For heavier hydrogen-like atoms it is closer to m. There are three ways to calculate the atomic mass, depending on the circumstances of each. Where subscripts $m_p$, e and n mean proton, electron and the neutron masses, respectively. In the hydrogen atom, in which the nucleus is just a proton, the ratio M / m is about 1836, so that 0.99946m. 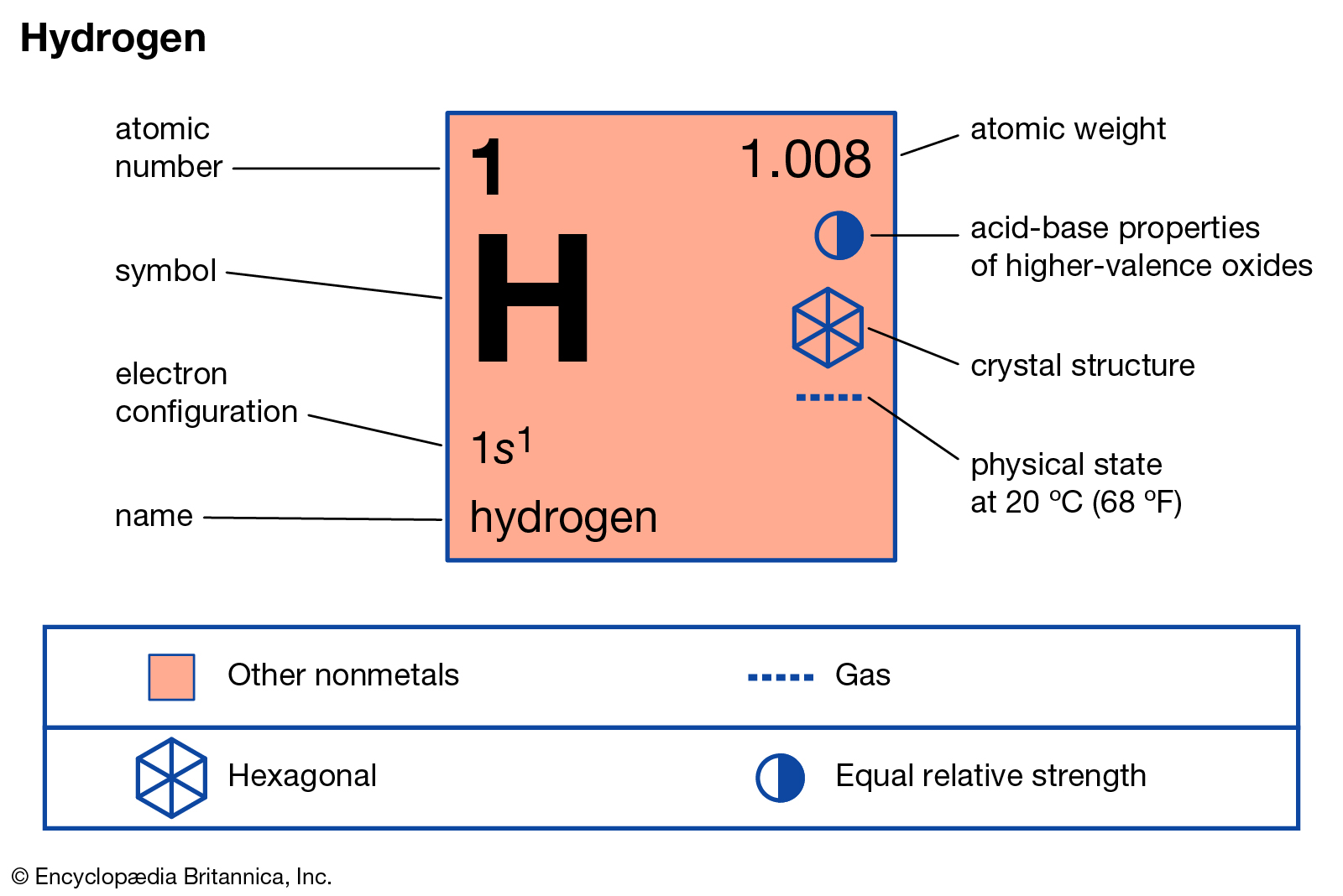 
Let me elaborate: the atomic mass of some nucleus $^A _Z X_N$ is defined as It appears that the mass of the simplest of the examples, hydrogen is not correctly produced, yet I cannot put my finger on what is the reason for this. After spinning around the atomic mass equation for calculating neutron separation energies, I have run into somewhat of a conundrum.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
